New Question Added to Help Determine Single IRB Requirements
We have added a new question to the Standard Proposal Questionnaire in Proposal Development that asks:
“Does this research proposal include domestic external sites that will be engaged in conducting non-exempt human research?”
This question was added to help determine if a project will require the use of a Single IRB, rather than each site obtaining separate IRB approvals from each of their institutions. For federal proposals, if you have answered “yes” to this question please contact MSU HRPP, IRBReliance@ora.msu.edu, as soon as possible. Some agencies, such as NIH, require a Single IRB Plan to be included with the proposal, which requires time and assistance from MSU HRPP. More information can be found by clicking on the More Information link in KC.
This question was added as part of the human subjects hierarchy, which means it will only appear if you have answered “yes” to the human subject’s question.
Does this research proposal include domestic external sites that will be engaged in conducting non-exempt human research?
10134: Does this research proposal include domestic external sites that will be engaged in conducting non-exempt human research?
Explanation: Human research studies that do not qualify for exemption that engage domestic external institutions in the conduct of the research may require use of a Single IRB. This means that rather than each site obtaining separate IRB approvals from each of their institutions, the sites may be required to rely upon a Single IRB that has been designated and agreed to by the sites. At MSU, the MSU Human Research Protection Program (HRPP) must agree to selection of the Single IRB. For NIH proposals, selection of the Single IRB occurs at the time of proposal submission. Contact the MSU HRPP early and as soon as possible if your study may involve a Single IRB. More information can be found on the MSU HRPP website: hrpp.msu.edu.
Regulation: For the NIH Single IRB requirement, see the Final NIH Policy on the Use of a Single Institutional Review Board for Multi-Site Research. For the Revised Common Rule requirement, see 45 CFR 46.114 (2018 Requirements).
Policy: For the NIH Single IRB requirement, see HRPP Manual Section 2-2-F-iii, Use of a Single IRB (U.S. Department of Health and Human Services, National Institutes of Health). For reliance requirements, see HRPP Manual Sections 1-3, Use of Institutional Authorization Agreements, 1-4, Reliance on External Independent (Commercial) Institutional Review Boards, and 1-5, Use of the National Cancer Institution Central Institutional Review Board. Visit hrpp.msu.edu for the HRPP Manual.
Yes
No
Proposal Development (PD) documents that were In Progress before the question was added may receive the following notice at the top of the Questionnaire tab:
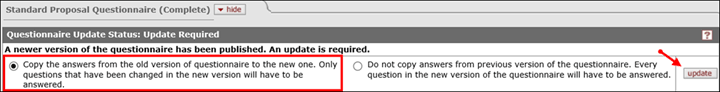
Leave the default answer selected and click the update button. This will bring in the new question, which will only be visible if you marked “yes” to the human subject’s question.
PD documents that were already in route when the question was added will not be impacted by the change to the Questionnaire.
Affiliate Web Request Form
We have added a new web request form on the SPA website for adding MSU Affiliates. If you need to set someone up as an affiliate, please use the new “Adding MSU Affiliates” web form to make your request.
For assistance with accessing the web forms, see Issue 48 of the System Implementation Updates.
As a reminder, affiliate requests are typically made for future MSU faculty and pre-doctoral fellowships being submitted System-to-System.
How to Add Fellows in KR
(Article revised on 4/27/22 for transition to Kuali Research)
Recently we’ve experienced some confusion on how to enter fellows in KR. The answer depends on the following scenarios:
- If the proposal is being submitted System-to-System and the sponsor requires the fellow to be listed as the PI on the application, then the fellow would need to be listed as the PI in KR. PI’s are automatically included in the route in KR, therefore if the fellow is a graduate student they would need to go through the affiliation process to access KR and approve the PD document. If the fellow is a post-doc at MSU they should already have access to KR. In these scenarios the mentoring faculty member is typically included as a Key Person.
- For all other fellowship applications the mentoring faculty member should be included as the PI, and the fellow should be included as a Key Person in KR. The fellow should not be added to the Credit Allocation.
If you have questions related to fellowship applications please contact your OSP Proposal Team.